
k (b) In 1988 three teams of scientists found that the Shroud of Turin, which was reputed to be the burial cloth of Jesus, contained about 91 percent of the amount of carbon-14 contained in freshly made cloth. 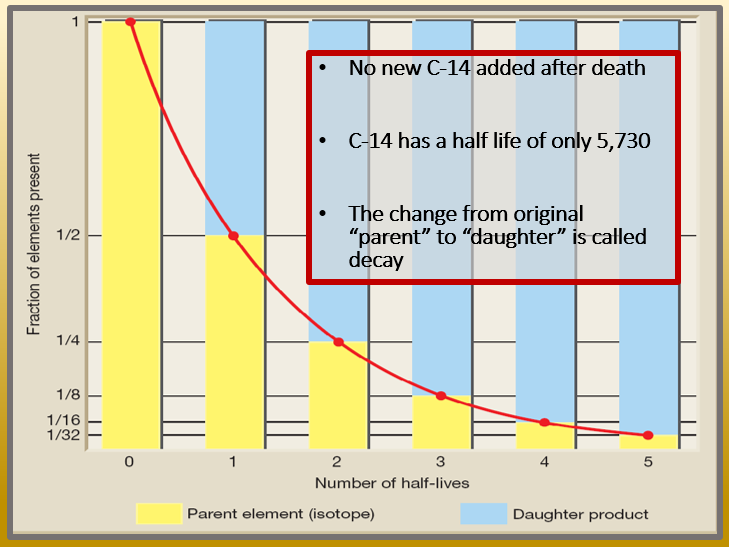
(a) Find the value of the constant k in the differential equation CkC. The following equation gives the quantitative relationship between the original number of nuclei present at time zero (N_0\) and the number (\(N\)) at a later time \(t\). Suppose C(t) is the amount of carbon-14 present at time t. Suppose C (t) is tho amount of carbon-14 present at time t. It is also applicable to the decay of excited states in atoms and nuclei. Question: The radioactive isotope carbon-14 is present in small quantities in all life forms, and it is constantly replenished until the organism dies, after which it decays to stable carbon-12 at a rate proportional to the amount of carbon-14 present, with a half-life of 5579 years. The concept of half-life is applicable to other subatomic particles, as will be discussed in Particle Physics. Nuclides with the shortest half-lives are those for which the nuclear forces are least attractive, an indication of the extent to which the nuclear force can depend on the particular combination of neutrons and protons. \)y for the least unstable, or about 46 orders of magnitude.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
